Content #1
Content #1
Content #1
Tyramide signal amplification (TSA) is a highly sensitive method that enables the detection of low-abundance targets in immunofluorescence. The technique works through enzymatic signal amplification: after primary antibodies (or biotinylated primary antibodies) bind to their targets, an HRP-conjugated secondary antibody (or streptavidin) catalyzes the covalent coupling of labeled tyramide to tyrosine residues on nearby proteins. This enzymatic coupling reaction deposits multiple fluorescent molecules at each target site, generating significantly stronger signal than conventional labeled secondary antibodies. TSA delivers exceptional sensitivity and signal-to-background ratios, making it possible to visualize low-abundance targets that cannot be efficiently detected using traditional immunostaining methods. Additionally, TSA compensates for the reduced antibody binding efficiency inherent to formalin-fixed, paraffin embedded (FFPE) samples, improving detection sensitivity of challenging targets in highly cross-linked tissues.
Biotium’s TyraMax™ Amplification Dyes are high-performance tyramide amplification dyes engineered to maximize the advantages of TSA while enabling robust multiplex imaging. These dyes provide bright, photostable signal with low background and exceptional chemical stability. Unlike other TSA dyes, TyraMax™ Dyes remain stable in TSA amplification buffer for up to 24 hours, making them ideal for automated staining workflows. With the widest selection of dye colors on the market, from blue to near-IR, TyraMax™ Dyes integrate seamlessly into standard multi-color panels for both conventional and spectral imaging. Together, these performance characteristics provide a reliable solution for sensitive detection and streamlined multiplexing using TSA.
In the sections below, we highlight example TSA workflows using TyraMax™ Dyes and multiplexing approaches that demonstrate how these reagents can be configured to achieve high sensitivity and flexible panel design. These workflows are not specific to TyraMax™ Dyes and can be adopted for other TSA dyes as well.
There are three common strategies for using TSA in multi-color fluorescence imaging:
A. Use TSA in combination with dye-conjugated antibodies or other immunostaining reagents.
B. Perform multiple rounds of TSA sequentially, performing a peroxidase quenching step after each round of labeling to inactivate the secondary antibody from the previous labeling step.
C. Perform multiple rounds of TSA sequentially, performing an antigen retrieval step after each round of labeling to remove all antibodies, while the dye or biotin that is covalently attached to the sample is retained. In this way, primary antibodies from the same species can be used.
In the next three sections we will discuss each of these strategies, followed by general recommendations for multi-color imaging with TSA.
Our TyraMax™ Dyes enable robust, multi-color fluorescence imaging and are compatible with dye-labeled antibodies and various cell staining methods (see Figure 1). In this section we provide a sample protocol for integrating TSA with conventional immunostaining techniques.
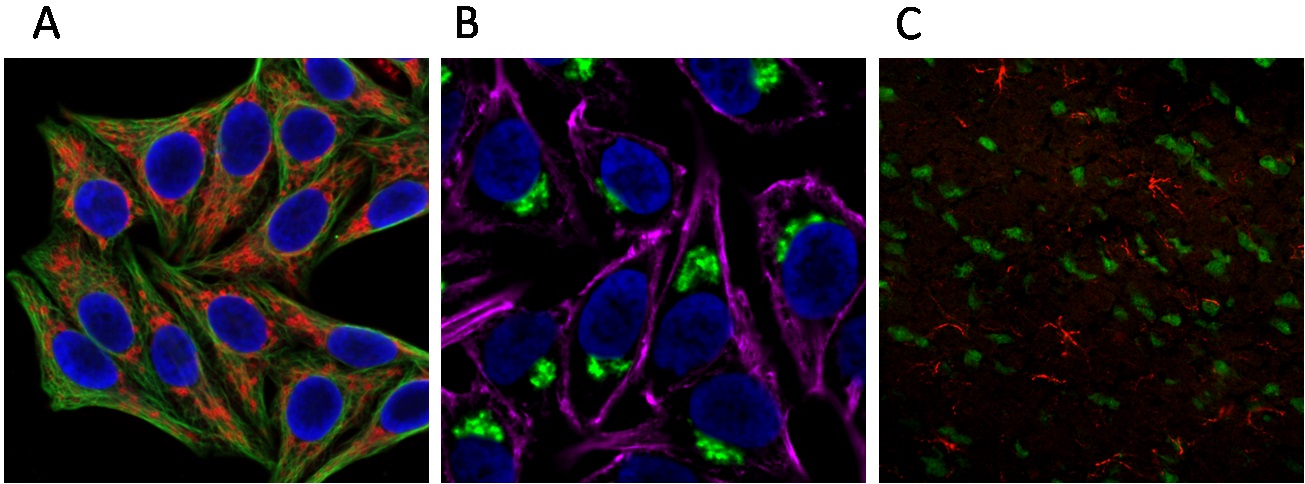
Figure 1. Multi-color imaging using TSA integrated into conventional immunofluorescence. (A) HeLa cells fixed with methanol; tubulin (green) detected with mouse anti-tubulin primary antibody and CF®488A Goat anti-mouse secondary antibody; mitochondria (red) visualized with rabbit anti-COXIV primary antibody and the Tyramide Amplification Kit with HRP Goat anti-rabbit IgG and CF®568 Tyramide (Cat. No. 33007); cell nuclei (blue) stained with DAPI. (B) HeLa cells fixed with formaldehyde; actin (magenta) detected with CF®640R phalloidin; Golgi bodies (green) visualized with mouse anti-Golgin-97 primary antibody and the Tyramide Amplification Kit with HRP Goat anti-mouse IgG and CF®488A Tyramide (Cat. No. 33000); cell nuclei (blue) stained with DAPI. (C) Rat brain section fixed with methanol; neuronal nuclei (green) labeled with CF®488A mouse anti-neuronal nuclei primary antibody; astrocytes (red) visualized with rabbit anti-GFAP primary antibody and Tyramide Amplification Kit with HRP Goat anti-rabbit IgG and CF®568 Tyramide (Cat. No. 33007).
Multiple rounds of TSA can be performed sequentially to label different targets within a single sample using primary antibodies (including biotinylated primary antibodies) from different host species. Following primary antibody labeling, the first target is detected using an HRP-conjugated secondary antibody (or streptavidin) and a TyraMax™ Dye. A peroxidase quenching step is then required to deactivate the HRP on the secondary antibody before proceeding to another round of TSA using a different color amplification dye.
Figure 2 illustrates multi-color imaging of a tissue section sequentially labeled using this peroxidase quenching-based approach. All probes provide strong fluorescence signals with minimal background and no detectable crosstalk. In this section, we provide a protocol for performing multiplex TSA using peroxidase quenching, which can be readily combined with other cell staining methods to achieve multi-color imaging.

Figure 2. Sequential labeling using TSA with peroxidase quenching. PFA-fixed rat eye cryosection stained with Vimentin Recombinant Monoclonal Mouse Antibody (rV9) HRP conjugate with TyraMax™ 630/650 (magenta; Cat. No. 96140) followed by peroxidase quenching, GFAP Recombinant Monoclonal Mouse Antibody (rGA5) HRP conjugate with TyraMax™ 560/580 (red; Cat. No. 96139), and CF®488A PSD95 Recombinant Monoclonal Mouse Antibody (rK28/43) (green). Nuclei are stained with NucSpot® 750/780 (cyan; Cat. No. 41038).
Antigen retrieval–also referred to as heat-induced epitope retrieval (HIER)–is commonly used to treat paraffin-embedded tissue sections in order to expose the epitopes that become cross-linked during fixation. It also strips away all antibodies and other reagents that bind to the sample via non-covalent interactions. By performing an antigen retrieval step after tyramide labeling, all antibodies (including primary antibodies and HRP-conjugated secondary antibodies) from the previous detection round are removed (antibody stripping), leaving only the covalently bound TyraMax™ Dyes. As a result, there’s no chance of cross-reactivity even when using multiple primary antibodies from the same host species, and all targets can be visualized simultaneously after multiple rounds of TSA with different color amplification dyes.
Various devices and protocols can be used for antigen retrieval, and sometimes they need to be specially optimized for certain tissue sections and/or targets. Here, we describe a technique based on microwave treatment (MWT) that is easy, fast, and effective for most of our samples, including both paraffin-embedded and frozen tissue sections (Figure 3).
Note: Microwaving is not recommended for cells in plastic plates or dishes, which may melt when heated.
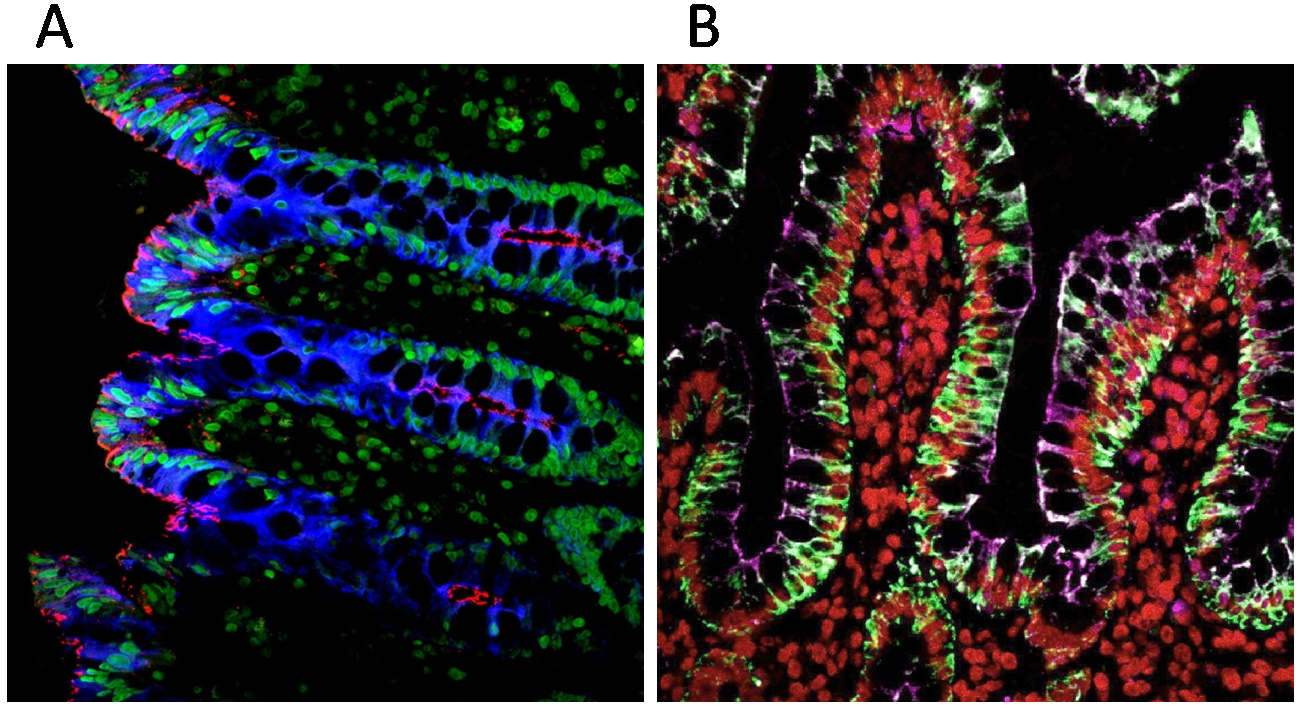
Figure 3. Antigen retrieval-based sequential labeling of cytokeratin (pan-CK) and tight junctions (ZO1). (A) Paraffin-embedded human colon section sequentially labeled with two CF® Dye Tyramides: the first round with HRP Goat Anti-Mouse IgG and CF®405S Tyramide, staining cytokeratin in blue, followed by antibody removal; the second round with HRP Goat Anti-Mouse IgG and CF®568 Tyramide, staining the tight junctions in red. The nuclei were stained with NucSpot® 470 nuclear stain (Cat. No. 40083, shown in green). (B) Paraffin-embedded human colon section sequentially labeled with three CF® Dye Tyramides: the first round with HRP Goat Anti-Mouse IgG and CF®488A Tyramide staining of cytokeratin in green; the second round with HRP Goat Anti-Mouse IgG and CF®640R Tyramide staining of tight junctions in magenta; the third round with HRP Goat Anti-Mouse IgG and Cyanine 555 Tyramide staining of histone H1 in red, with antibody removal performed between each round.
Among the three strategies discussed above, the antigen retrieval method offers the best signal-to-noise and the most flexible antibody selection without any issue of cross-talk. However, the microwave treatment or other antigen retrieval techniques may or may not be compatible with all applications. For example, chambered cover glass commonly used in cell imaging may be damaged during microwave treatment. Which multiplexing strategy to use should be determined experimentally based on the nature of samples and targets. The following technical tips highlight key considerations for selecting and implementing TSA-based multiplexing strategies.
Biotium offers a wide selection of reagents for TSA available as complete kits or individual components, providing the flexibility to tailor amplification strategies to specific experimental needs while maintaining consistent, high-quality performance. These reagents can be used individually or combined into complete TSA workflows, readily integrating into existing immunofluorescence protocols. Biotium’s TSA products support diverse experimental designs, from straightforward signal enhancement of a single low-abundance target to highly multiplexed panels using sequential tyramide labeling.
| Dye | Abs/Em (nm) | Laser Line | Detection channel | Dye Features | Size | Catalog No. |
|---|---|---|---|---|---|---|
| TyraMax™ 410/450 | 408/452 | 405 nm | DAPI/Alexa Fluor® 405 | Unique tyramide color, spectrally similar to Alexa Fluor® 405 | 20 uL | 96134-20UL |
| 100 uL | 96134-100UL | |||||
| TyraMax™ 430/500 | 421/497 | 405 nm | FITC | Brighter than Aluora® 430 and Opal® 480 | 20 uL | 96135-20UL |
| 100 uL | 96135-100UL | |||||
| TyraMax™ 400/550 | 394/547 | 405 nm | FITC | Unique tyramide color, spectrally similar to Pacific Orange® | 20 uL | 96136-20UL |
| 100 uL | 96136-100UL | |||||
| TyraMax™ 490/520 | 497/518 | 488 nm | FITC | Brighter than Opal® 520, replacement for Aluora® 488 | 20 uL | 96137-20UL |
| 100 uL | 96137-100UL | |||||
| TyraMax™ 555/565 | 552/569 | 555 nm or 561 nm | TRITC | Brighter than Opal® 570, replacement for Aluora® 555 | 20 uL | 96138-20UL |
| 100 uL | 96138-100UL | |||||
| TyraMax™ 560/580 | 562/584 | 555 nm or 561 nm | TRITC | Alternative to Aluora® 555, Opal® 570 with superior photostability | 20 uL | 96139-20UL |
| 100 uL | 96139-100UL | |||||
| TyraMax™ 630/650 | 631/650 | 633 nm or 640 nm | Cy®5 | Bright and photostable alternative to Aluora® 647, Opal® 650 | 20 uL | 96140-20UL |
| 100 uL | 96140-100UL | |||||
| TyraMax™ 647/670 | 650/670 | 633 nm or 640 nm | Cy®5 | Brighter than Opal® 650, replacement for Aluora® 647 | 20 uL | 96141-20UL |
| 100 uL | 96141-100UL | |||||
| TyraMax™ 660/680 | 663/683 | 633 nm or 640 nm | Alexa Fluor® 680 | Unique tyramide color, spectrally similar to Alexa Fluor® 660. Brighter and more photostable Opal® 690 when excited at 640 nm | 20 uL | 96142-20UL |
| 100 uL | 96142-100UL | |||||
| TyraMax™ 680/700 | 680/701 | 685 nm (detectable with 640 nm excitation) | Alexa Fluor® 680 | Brighter and more photostable than Opal® 690 when excited at 685 nm | 20 uL | 96143-20UL |
| 100 uL | 96143-100UL | |||||
| TyraMax™ 710/740 | 711/736 | 685 nm | Alexa Fluor® 700 | Brighter and more photostable than Aluora® 700 | 20 uL | 96144-20UL |
| 100 uL | 96144-100UL | |||||
| TyraMax™ 740/770 | 742/768 | 730 nm | Alexa Fluor® 750 | Single-step detection, unlike Opal® 780. Stable in amplification buffer for up to 24 hours, unlike Aluora® 750 | 20 uL | 96145-20UL |
| 100 uL | 96145-100UL |
| Tyramide Label | Ex/Em | Secondary conjugate | Catalog no. |
|---|---|---|---|
| CF®488A | 490/515 nm | Goat anti-mouse HRP | 33000 |
| Goat anti-rabbit HRP | 33001 | ||
| Streptavidin HRP | 33002 | ||
| CF®543 | 541/560 nm | Goat anti-mouse HRP | 33003 |
| Goat anti-rabbit HRP | 33004 | ||
| Streptavidin HRP | 33005 | ||
| CF®568 | 562/583 nm | Goat anti-mouse HRP | 33006 |
| Goat anti-rabbit HRP | 33007 | ||
| Streptavidin HRP | 33008 | ||
| CF®594 | 593/614 nm | Goat anti-mouse HRP | 33009 |
| Goat anti-rabbit HRP | 33010 | ||
| Streptavidin HRP | 33011 | ||
| CF®640R | 642/662 nm | Goat anti-mouse HRP | 33012 |
| Goat anti-rabbit HRP | 33013 | ||
| Streptavidin HRP | 33014 | ||
| CF®680R | 680/701 nm | Goat anti-mouse HRP | 33015 |
| Goat anti-rabbit HRP | 33016 | ||
| Streptavidin HRP | 33017 | ||
| Biotin-XX | N/A | Goat anti-mouse HRP | 33018 |
| Goat anti-rabbit HRP | 33019 | ||
| Streptavidin HRP | 33020 |
| Tyramide label | Ex/Em | Size | Catalog no. |
|---|---|---|---|
| CF®350 | 347/448 nm | 0.5 mg | 92170 |
| CF®405L | 395/545 nm | 0.5 mg | 92198 |
| CF®405S | 404/431 nm | 0.5 mg | 92197 |
| CF®405M | 408/452 nm | 0.5 mg | 96057 |
| CF®430 | 426/498 nm | 0.5 mg | 96053 |
| CF®488A | 490/515 nm | 0.5 mg | 92171 |
| FITC | 492/514 nm | 0.5 mg | 96018 |
| CF®514 | 516/548 nm | 0.5 mg | 92199 |
| CF®532 | 527/558 nm | 0.5 mg | 96066 |
| CF®543 | 541/560 nm | 0.5 mg | 92172 |
| CF®550R | 551/577 nm | 0.5 mg | 96077 |
| CF®555 | 555/565 nm | 0.5 mg | 96021 |
| Cyanine 555 | 555/565 nm | 0.5 mg | 96020 |
| CF®568 | 562/583 nm | 0.5 mg | 92173 |
| CF®583R | 586/609 nm | 0.5 mg | 96085 |
| CF®594 | 593/614 nm | 0.5 mg | 92174 |
| CF®620R | 617/639 nm | 0.5 mg | 92194 |
| CF®640R | 642/662 nm | 0.5 mg | 92175 |
| CF®647 | 650/665 nm | 0.5 mg | 96022 |
| CF®660R | 663/682 nm | 0.5 mg | 92195 |
| CF®680R | 680/701 nm | 0.5 mg | 92196 |
| CF®710 | 712/736 nm | 0.5 mg | 96127 |
| CF®725 | 729/755 nm | 0.5 mg | 96128 |
| CF®740 | 742/767 nm | 0.5 mg | 96124 |
| CF®750 | 755/779 nm | 0.5 mg | 96052 |
| CF®754 | 748/793 nm | 0.5 mg | 96090 |
| Biotin-XX | N/A | 0.5 mg | 92176 |
| DNP | N/A | 0.5 mg | 96019 |